Your new post is loading...

|
Scooped by
Beeyond
|
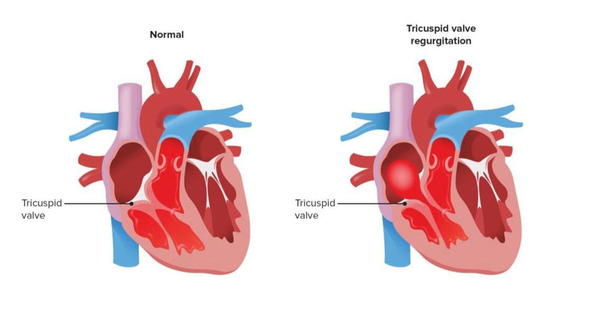
The heart, is a vital organ beating rhythmically, fuels life throughout our bodies. However, for millions worldwide, heart valve diseases disrupt this vital rhythm. Fortunately, recent years have witnessed groundbreaking progress in heart valve replacement surgery in India, offering renewed hope and an enhanced quality of life for those in need. Raising awareness about these advancements is crucial for optimal utilization and patient benefit.

|
Scooped by
Beeyond
|
MVR performed with the On-X valve is an attractive surgical option for patients with mitral valve IE, with its associated mortality and morbidity rates being comparable to those associated with the use of the SJM valve plus its superior hemodynamic performance. EOA appears to be of independent predictive value for the occurrence of late-new-onset AF in patients who have undergone MVR for IE. The EOA achieved with the use of the On-X valve in the mitral position may reduce the incidence of new-onset AF in the late phase.

|
Scooped by
Beeyond
|
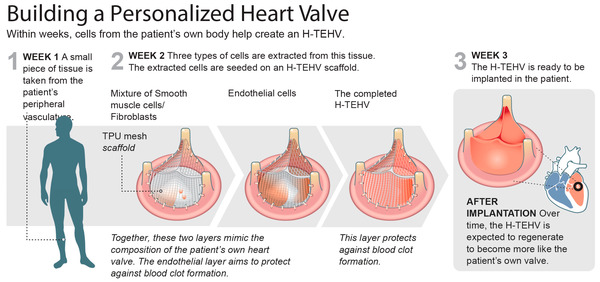
We have successfully developed, tested, and, for the first time, implanted a novel H-TEHV featuring a permanent elastomeric scaffold in sheep. Our initial findings suggest that the H-TEHV demonstrates resilience under high-pressure conditions at the mitral position, exhibiting no signs of shrinkage or valve decomposition. While these short-term results may suggest the feasibility of the H-TEHV concept in vivo, it is crucial to acknowledge that given the current stage of development and the limited scope of a single animal implant, no definitive conclusions can be drawn regarding the long-term suitability of the H-TEHV.

|
Scooped by
Beeyond
|
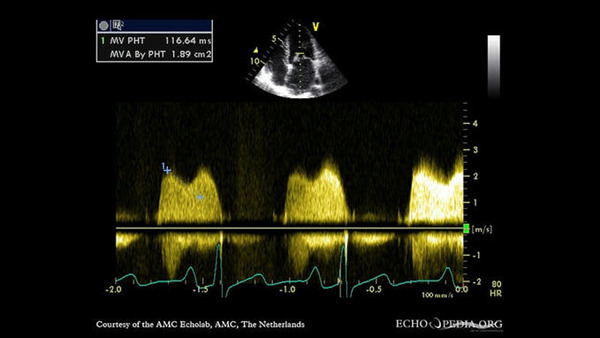
A novel technology called intravascular lithotripsy-facilitated percutaneous balloon mitral valvuloplasty (IVL-PBMV) shows promise for treating patients with severe calcific mitral stenosis (MS) and no other surgical or transcatheter treatment options. But until recently, evidence of its value has been sparse, consisting mainly of isolated case reports from various centers.

|
Scooped by
Beeyond
|
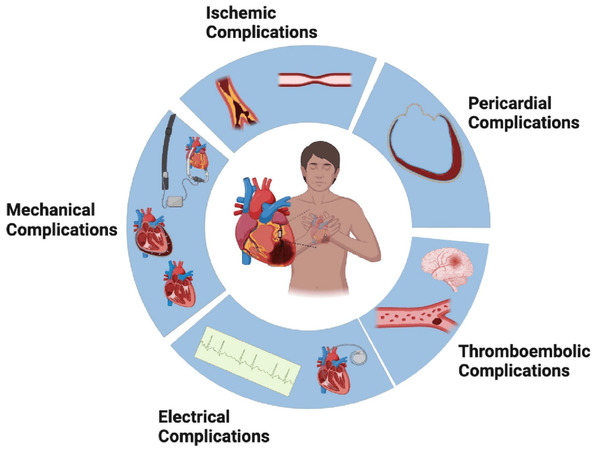
The complications of AMI represent high-acuity, time-sensitive conditions associated with elevated morbidity and mortality rates. Early recognition and decisive intervention remain the cornerstones in the quest for optimal patient outcomes. Given the dramatic presentations associated with complications and the urgent need for intervention, early revascularization has become the standard of care, resulting in a reduced incidence of complications of less than 0.1%. Furthermore, emphasizing patient-centered planning and the judicious timing of appropriate interventions, including surgical procedures, percutaneous technologies, and mechanical circulatory support involvement, holds the potential to significantly enhance both disease- and patient-centered outcomes (Table 1). As a result, a multidisciplinary heart team emerges as a crucial entity in guiding the care of patients post-AMI with complications.

|
Scooped by
Beeyond
|
The following is a summary of “Valve unit instead of intensive or intermediate care unit admission following transcatheter edge-to-edge mitral valve repair is safe and reduces postprocedural complications,” published in the February 2024 issue of Cardiology by Gröger et al.

|
Scooped by
Beeyond
|
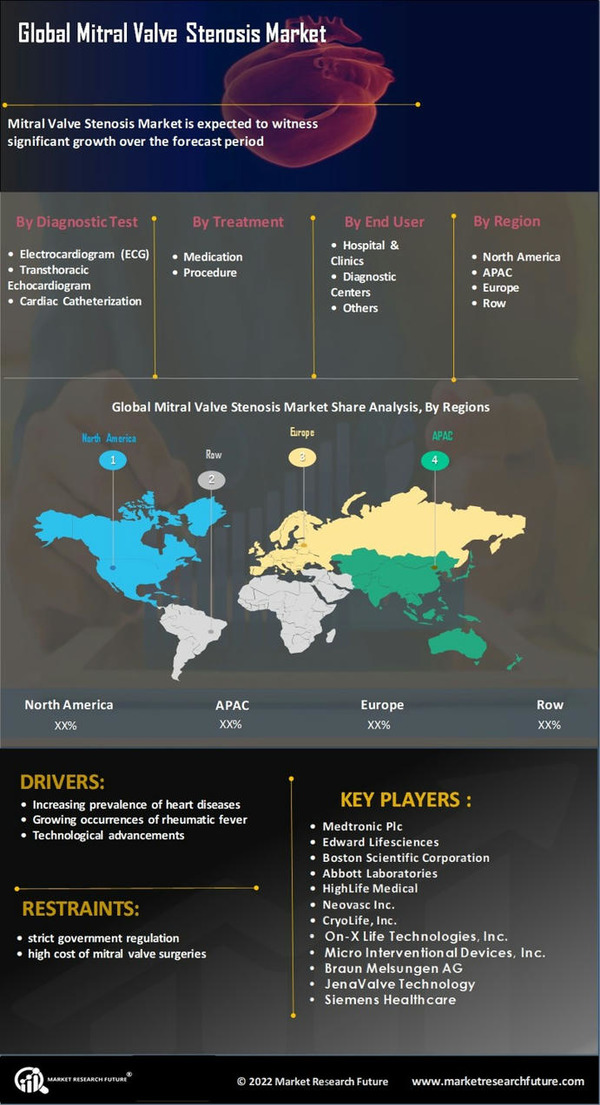
The global mitral valve stenosis market is expanding over time, owing to reasons such as rising heart disease prevalence, rising rheumatic fever cases, technical improvements, the launch of innovative mitral valves, Cardiac Valve, and an expanding number of significant industry players. North and South America are expected to dominate the global mitral valve stenosis market thanks to their high adoption of innovative medical devices, rising healthcare expenditure, and increased occurrence of heart diseases.

|
Scooped by
Beeyond
|
Tissue engineering aims to overcome the current limitations of heart valves by providing a viable alternative using living tissue. Nevertheless, the valves constructed from either decellularized xenogeneic or purely biologic scaffolds are unable to withstand the hemodynamic loads, particularly in the left ventricle. To address this, we have been developing a hybrid tissue-engineered heart valve (H-TEHV) concept consisting of a nondegradable elastomeric scaffold enclosed in a valve-like living tissue constructed from autologous cells.

|
Scooped by
Beeyond
|
A less-traumatic alternative to the traditional open-heart surgery, minimally-invasive structural heart interventions have made significant advances in the last few years. Interestingly, there is a wide range of cardiac diseases -- such as valvular heart disease, congenital defects and heart failure -- that can be treated with this approach. Dr Haresh G Mehta, director-interventional cardiology and Dr Kayan Siodia, consultant interventional cardiology, S L Raheja Hospital, Mahim -- a Fortis associate say that for patients with valvular heart disease, valve replacement or repair through open heart surgery was the only option, which had a "high procedural risk" and longer recovery time.

|
Scooped by
Beeyond
|
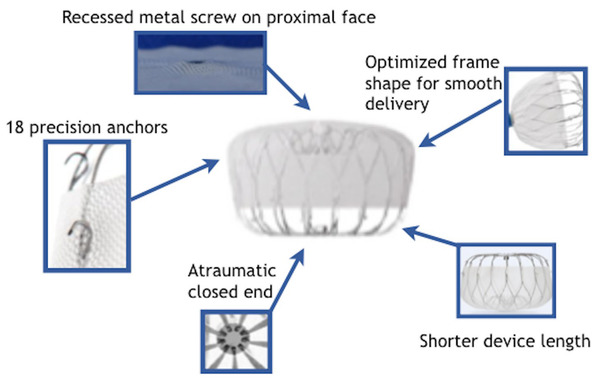
As the first self-expanding TPVR product approved in China and Europe, VenusP-Valve carries remarkable clinical value. Uniquely designed with both flared ends, the product ensures the blood flow of branchial artery with bare stents at the outflow end. It provides a stable multi-point anchoring system and enables easy delivery, with no need for pre-stenting before the procedure. Available in a variety of specifications with extensive applicability, VenusP-Valve is able to meet the needs of 85% of patients in the case of large RVOT.

|
Scooped by
Beeyond
|
Left atrial appendage occlusion (LAAO) is an increasingly used alternative to oral anticoagulation in patients with atrial fibrillation, especially in patients with absolute/relative contraindications to these therapies. This review will cover three main aspects of the procedure. In the fist part of the manuscript, we focus on patient selection. We describe three main categories of patients with primary indication to LAAO, namely patients with previous or at a high risk of intracerebral bleeding, patients with a history of major gastrointestinal bleeding and patients with end-stage renal disease and absolute contraindication to novel oral anticoagulants.

|
Scooped by
Beeyond
|
Tampa General Hospital is one of just five health care systems across the nation approved by the Food and Drug Administration (FDA) to participate in a feasibility study using the AltaValve technology for TMVR. As an academic health system, Tampa General and USF Health combine clinical care, academics and research to deliver the best possible patient outcomes. This robust research enterprise enables providers to use cutting-edge technologies, resources and therapies to address some of the most complex health conditions.

|
Scooped by
Beeyond
|
Climate change-related CV burden represents one of the latest challenges in preventive cardiology that physicians are facing. Although the impact of climate change on human health remains to be accurately estimated, several studies have demonstrated its negative effects on the risk and progression of CV diseases. Therefore, serious strategies should be planned to contrast global warming and climate change, involving physicians, researchers, public health professionals, political scientists, legislators, and national leaders who have to work together to alleviate the impacts of climate change and preserve public health.

|
Scooped by
Beeyond
|
ShortCut leaflet modification device offers a safe, simple and effective way to split valve leaflets and ease valve-in-valve Transcatheter Aortic Valve Replacement procedures in patients who are at risk of coronary obstruction

|
Scooped by
Beeyond
|
The PRIMARY trial is directed by Joanna Chikwe, MD, chair of the Department of Cardiac Surgery at Cedars-Sinai. The randomized trial--across 60 sites in six countries--compares transcatheter edge-to-edge mitral repair (using a clip) to surgical repair for patients with severe mitral regurgitation caused by mitral valve prolapse.

|
Scooped by
Beeyond
|
Patients were followed regularly after the procedure through clinic visits during which transthoracic echocardiography and contrast CT were done unless contraindicated (90% also had at least one cardiac CT, and 79% had at least one TEE during follow-up).

|
Scooped by
Beeyond
|
Testing various combinations of Viabahn covered stents and Amplatzer vascular plugs, researchers found that all device sizes resulted in immediate elimination of target PVL associated with transcatheter heart valve (THV) implants in eight patients. There were no intraprocedural complications for the approach, dubbed the "Tootsie Roll" technique by Vasilis Babaliaros, MD, of Emory University Hospital Midtown in Atlanta, and colleagues.

|
Scooped by
Beeyond
|
The authors of the paper found in total that 39% of patients had postoperative AF after surgery, and almost one-quarter of those patients developed new or recurrent AF after 30 days. Almost 5% of patients with postoperative AF experienced a neurologic event, such as stroke, the researchers report.

|
Scooped by
Beeyond
|
The Tria mitral valve is designed specifically for the anatomy and pressures of the mitral position. It combines the company’s proprietary polymer—LifePolymer—with an innovative valve design intended to resist calcification, withstand stresses and strains without failure, and restore patient quality of life without lifelong use of anticoagulants.

|
Scooped by
Beeyond
|
“Our approach to TMVR is unique in that we work with the natural movement of the heart, as opposed to first-generation technologies which require the heart to work around a stiff implant. Our flexibility and four-point securement system provide a patent LVOT and maintains left ventricular function throughout recovery,” said Julie Logan Sands, founder and CEO ReValve Solutions.

|
Scooped by
Beeyond
|
Research has been conflicting on whether patients would benefit from more aggressive early postoperative anticoagulation treatment, the researchers claim. Their study compared all-cause mortality and thromboembolic events in patients undergoing surgical aortic valve replacement (SAVR) receiving anticoagulation with warfarin versus patients with no systemic anticoagulation.

|
Scooped by
Beeyond
|
The ARISE II study is the first multicentre pivotal study approved by the US Food and Drug Administration (FDA) investigating the use of a minimally invasive endovascular device to treat the ascending aorta. The study investigates the treatment of isolated lesions as well as chronic and residual type A dissections involving the ascending aorta. The Gore ascending stent graft is designed for investigational use in combination with the Gore Tag thoracic branch endoprosthesis.

|
Scooped by
Beeyond
|
The CardioMech device is an artificial chord that is designed to reduce or eliminate regurgitation and to restore the native anatomy. The catheter-based therapy is intended to be a simple and straightforward procedure and an alternative therapy option for both patients eligible for open-heart surgery and those considered surgically ineligible.

|
Scooped by
Beeyond
|
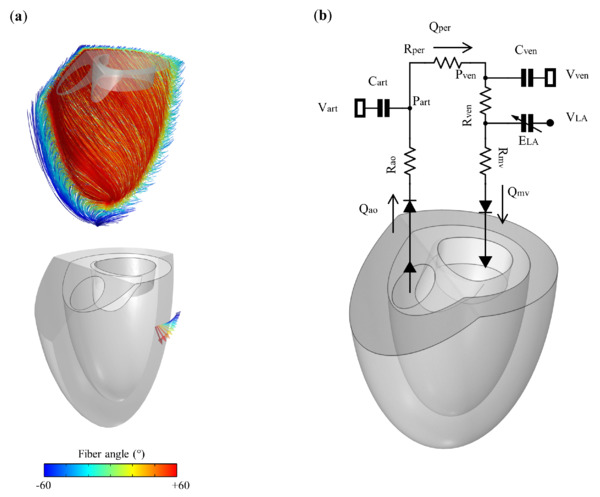
The 3D-0D computational model presented in this study offers valuable insights into the flow patterns within the heart under various conditions, such as different angles of MV anterior leaflet angulation and MV laceration, in the presence of HCM. The utilization of simplified geometric models for the LV and MV decreases the computational workload, hence enhancing the process of model creation. The passive component of ventricular myocardial tissues was assumed to exhibit transverse isotropic hyperelasticity, despite their orthotropic characteristics.

|
Scooped by
Beeyond
|
Medical device startup CardioMech raised $13 million to advance the development and validation of its flagship device, which is designed to treat degenerative mitral valve regurgitation. The catheter-based device seeks to offer an uncomplicated, direct procedure for patients eligible for open-heart surgery as well as those considered eligible for the surgery.
|

Curated by Beeyond
BEEYOND is a consulting company in the field of disruptive innovation, accompanying established companies on out-of-the-core growth strategy, from creation of new concepts to product launch. Reach us at: contact@beeyond.fr.
|

 Your new post is loading...
Your new post is loading...